 
This content was accessible as of December 29, 2012, and it was downloaded then by Andy Schmitz in an effort to preserve the availability of this book. 
#ELECTRON AND MOLECULAR GEOMETRY CALCULATOR LICENSE#See the license for more details, but that basically means you can share this book as long as you credit the author (but see below), don't make money from it, and do make it available to everyone else under the same terms. Based on the chart, the molecular geometry for BF 3 would be trigonal planar, with an angle of 120 degrees between the bonds.This book is licensed under a Creative Commons by-nc-sa 3.0 license. If we drew the electron dot structure for BF 3, boron trifluoride, we will notice that there are three attachment point, and 3 bonds, to the central atom, boron. What is the molecular geometry of BF 3, boron trifluoride? Now that we know the molecular geometry, we can determine the bond angle to be about 105 degrees from our chart. Therefore, the resulting molecular geometry is a a bent geometry. Two of these attachments are bonds and the other two are lone pairs. However, this is not the molecular geometry. This would make the electron geometry tetrahedral. Notice there are 4 attachments, or, electron groups surrounding oxygen. The answer is the molecular geometry of water would be bent. Practice Example: What is the molecular geometry and bond angle of water (H 2O)? For the most part, this information will have to be memorized. In the table below, you will see the coordination between the number and type of attachments in relation to the bond angles. Notice in the table below how if there are no lone pairs, the molecular geometry and electron geometry will be the same. additionally, we need to know how many of these attachments are bonds and lone pairs. Firstly, we must know how many total attachments there are. To determine the molecular geometry of a structure we need to know two things. Configurationĭetermining molecular geometry and bond angles Below is a table demonstrating the relationship between the number of bonding partners and these configurations. There are three main types of configurations: linear, trigonal, and tetrahedral. This theory revolves around the idea that electrons repel each other and therefore will bond accordingly. Chemists are able to predict the arrangement of atoms and chemical bonds using the valence-shell electron-pair repulsion theory or VSEPR. Molecular geometry describes the three-dimensional structure of a molecule. Bond angles: The angle between adjacent bonds of an atom. 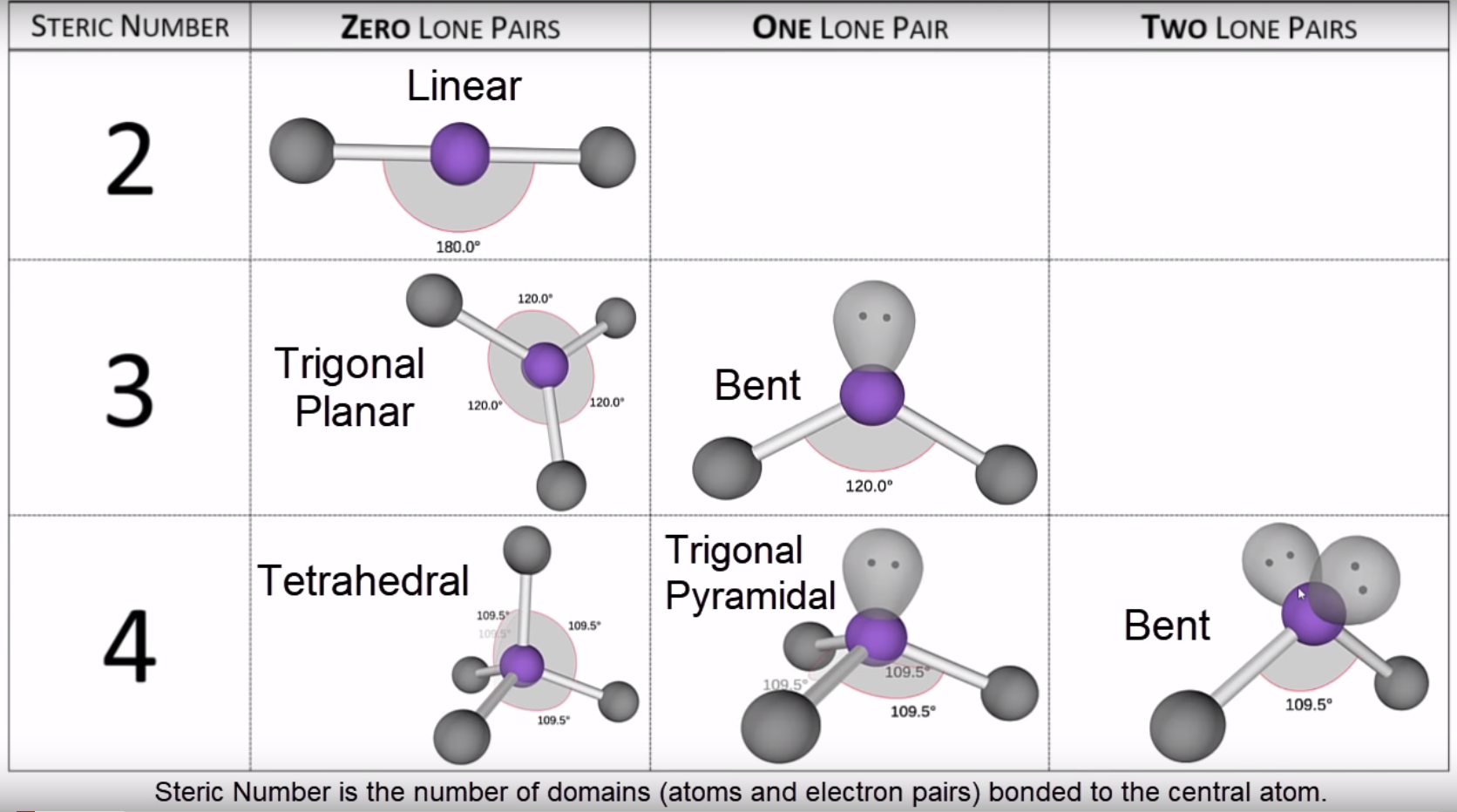 
Hybridization: Orbitals are combined in order to spread out electrons.Molecular Geometry: Describes the arrangement of atoms around the central atom with acknowledgment to only bonding electrons.Electron Geometry: Describes the arrangement of bonds and lone pairs around a central atom. #ELECTRON AND MOLECULAR GEOMETRY CALCULATOR FREE#If you enjoy this tutorial, feel free to check out our other tutorials on bonding listed below. You will learn about the more common molecular geometries: tetrahedral, linear, bent, trigonal pyramidal, and trigonal planar – along with their bond angles. #ELECTRON AND MOLECULAR GEOMETRY CALCULATOR HOW TO#In this tutorial, you will learn how to identify the molecular geometry and bond angles of a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
